Bioethics Forum Essay
Xenotransplantation: Three Areas of Concern
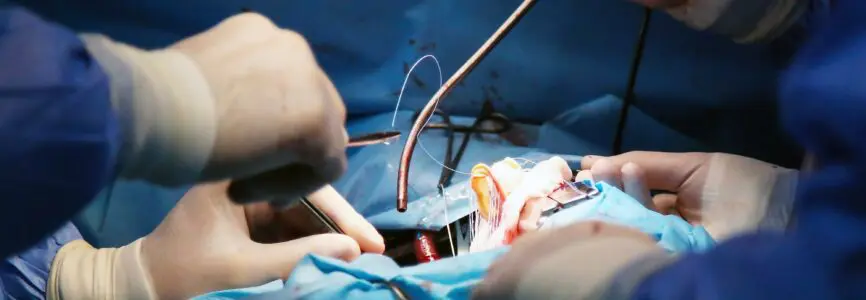
News of the first transplant of a pig’s heart into a human was widely reported last week, raising hope that the procedure could one day help alleviate the shortage of organs. In decades of experimentation with xenotransplantation, the transplantation of organs from one species to another, no human recipient of a nonhuman organ has survived for more than a few months. Most have died within hours or days from rejection, infection, or both. Like the recipient of the pig’s heart, many previous patients were dying and were not eligible for an organ transplant through conventional channels. But before we forge ahead with clinical trials of xenotransplantation we should be concerned about several issues: the potential to spread pathogens, exploitation of human research participants, and animal welfare.
Early research on xenotransplantation used organs from monkeys and chimpanzees, who are the closest phylogenetic and evolutionary relatives to humans. That research ended because of concerns about the transmission of pathogens, including retroviruses, some of which are readily transmitted from nonhuman primates to humans. The U.S. Food and Drug Administration effectively banned the use of nonhuman primates for xenotransplantation in 1999, citing the unacceptably high risk of zoonotic infection. In recent years, with the availability of genetic technologies like CRISPR-Cas-9, scientists have sought to genetically modify pigs to grow human-compatible organs for transplant. The genetic modifications are intended to reduce the risk of rejection and to inactivate or eliminate pathogens harbored in pig cells. The ultimate goal of this research is to turn pigs into a ready source of organs for transplant, as part of the effort to address the chronic shortage of organs. As scientist Claus Hammer has described it, for xenotransplantation to succeed, “we need to ‘outwit’ the 180 million years of evolution.”
We are in the third year of a global pandemic that has killed millions of people worldwide and sickened many millions more. SARS-CoV-2 is a zoonotic virus that has been identified in many wild, captive, and domestic animal species. The potential dangers of known and unknown pathogens unleashed through xenotransplantation are impossible to quantify or predict, but heightened concern is warranted.
Xenotransplantation remains experimental, and potential recipients of xenografts are research subjects whose consent and safety are governed by human subjects research regulations and guidance. Several regulatory and advisory organizations have concluded that it is imperative for xenotransplant recipients to submit to extended, possibly lifelong surveillance for zoonotic infectious diseases. The U.S. Public Health Service calls for lifelong surveillance of recipients to monitor for infectious agents. The Nuffield Council on Bioethics in the U.K. notes that the xenotransplant recipient, their close contacts, family, and sexual partners, and the transplant and health care teams should all be monitored for unexplained illness. “Patients consenting to xenotransplantation should be informed that post-operative monitoring for infectious organisms is an integral part of the procedure, and that their consent to the operation includes consent to this monitoring,” Nuffield states.
Requiring lifelong surveillance and submission to monitoring as a condition of research participation in xenotransplantation trials is in tension with the right of research subjects to withdraw from research at any time, for any reason. This right is endorsed worldwide in ethical guidelines governing human subjects research and is a critical component of voluntary informed consent. The U.S. Common Rule states that research “participation is voluntary . . . and the subject may discontinue participation at any time without penalty or loss of benefits.” Requiring lifelong surveillance effectively denies research subjects their fundamental right to withdraw, and violates international norms and ethical guidance. Surveillance and notification of close contacts impinge on the right to privacy and confidentiality of patients and research subjects.
There are also novel ethical concerns about the use of pigs to grow organs. While pigs are farmed and killed in the millions for meat, the pigs used for xenotransplantation are categorically different animals, and their use in research is governed by different welfare considerations. These pigs are genetically modified and cloned, and must be bred and housed using infection-control measures like artificial insemination, embryo transfer, Caesarian births, and isolation in sterile environments without contact with other animals, preventing the expression of their natural behaviors. Their use would require frequent blood and tissue sampling, which in pigs requires restraint, including drug-induced restraint. If pigs are used for multiple tissue and organ transplants, they could be subjected to repeated surgeries, causing these highly intelligent and social animals pain and distress. Additionally, although nonhuman primates have been ruled out as organ sources, they are still used in xenotransplantation research as experimental recipients of organs from pigs, raising similar welfare concerns. Using pigs and nonhuman primates for xenotransplantation research, or to grow organs, violates established best practices for animal care and welfare, which include providing ethologically appropriate environments that meet the animals’ behavioral and physiological needs.
The organ shortage is a formidable problem. More than 100,000 people in the U.S. are on waiting lists for organ and tissue transplants, and 17 people die each day while waiting. But using pigs to grow organs is only one of many possible solutions to the shortage. Access to basic health care and therapies to treat common diseases like hypertension, diabetes, and heart disease can prevent organ failure. Using expanded criteria organs, such as those from older donors and donors with chronic diseases, has already increased the supply of organs. Other promising solutions include the use of stem cells to grow human organs and possibly 3D bioprinting to repair organs. Low-tech social engineering, such as improving communication about organ donation, and implementing opt-out organ donor registration, should be used to encourage more people to become organ donors. At a time of growing public concern about animal welfare, a genuine commitment to reducing the numbers of animals killed for research and other purposes favors seeking out and developing alternatives. As the SARS-CoV-2 pandemic has demonstrated, the health and well-being of humans and animals are entangled. Zoonotic pandemics are a grave threat both to humanity and to other creatures, and a compelling reason to reevaluate the risks and reconsider xenotransplantation as a path forward.
L. Syd M Johnson, PhD, (@LSydMJohnson) is an associate professor at the Center for Bioethics and Humanities and a clinical ethics consultant at Upstate Medical University in Syracuse, N.Y.












Tipos de traducción
Traducción de texto
Texto original
1.203 / 5.000
Resultados de traducción
L. Syd M. Johnson notes reflections in three lines of great importance. He mentions, regarding the intimate surveillance to which those undergoing xenotransplantation will be subjected, the violations of Informed Consent and Good Clinical Practices and it is the least that those of us in Research Ethics Committees could demand of respect. The experience of this pandemic has wreaked havoc on many families due to the loss of loved ones, but also due to economic losses and family breakups caused by long isolations, so taking new risks for another zoonosis is not something that many of us want.
The border in this field is a very fine border that must keep the integrity of the subject safe. It would be worth asking if the person who receives a pig’s heart agrees to “give up” the rights to it in exchange for living a few more days. That I myself would respond to the question is idle, because only those who live those last moments over and over and over again while waiting for an organ can answer.
The reflections help us, the Ethics Committees, to give a voice to those who do not have a voice: the research subjects.
This article provided an insightful analysis into xenotransplantation and highlights important ethical concerns.
I am drawn to implications regarding informed consent and this article emphasized essential components that must be considered for the protection of patients. Reflecting on xenotransplantation and informed consent, the risk of therapeutic misconception is a primary consideration of mine. Xenotransplantation presents the possible benefit of extending longevity and quality of life for recipients, which may overshadow the risks to the procedure that this article highlighted. Thus, I question how the procedure will ensure patients are not unduly influenced based on the potential associated benefit.
The insightful analysis into the use of pigs as the supply for donor organs and associated ethical issues are another prominent concern that I feel as though researchers must address as well. Pigs have been cited as the most suitable donor animals due in part to their large litter sizes, short periods of maturation, and physiologic similarity to human organs, thus, they become subjected to significant genetic modification and testing for human benefit. With the continued development of xenotransplantation research, it may be in the interest of researchers and regulatory bodies to heavily monitor sites in order to ensure welfare standards are upheld as much as possible for these animals. The use of pigs ultimately raises ethical concerns, but the research community can work and advocate to minimize the level of harm imparted to such animals.
It is with hope that the recent case will provide valuable knowledge into the field of xenotransplantation and ultimately guidance for upholding the bioethical principles to protect both donor animals and recipients. Thank you for sharing this great article!
Our ever increasing need for organ donation is a pressing issue, and this article which tackles the topic of xenotransplantation (animals to humans) was thought provoking. The author brings up two main issues with xenotransplantation, first, the inability of participants to safely withdraw from the trials once involved, and secondly, the ethical implications of enrolling countless animals in these trials.
A key component of research is the ability of its participants to withdraw at any point in time, this effectively gives them autonomy over their own situation and prevents people from being coerced or forced into research trials. The author of this article highlights the issue with xenotransplantation trial participants, because of the risk of zoonotic disease involved with receiving an animal organ, participants must undergo lifelong surveillance to prevent disease from spreading through society. Especially in this day and age, where we are just now recovering from a global battle against Covid-19, legislation is extra sensitive to animal related research. Because of how important our research regarding xenotransplantation is, I believe we should waive these previously established standards in order to conduct these experiments.
I believe the second point the author brought up, the ethical implications of experimenting on animals, highlights a dichotomy in societal views of respect towards animals. The majority of people across the globe have turned a blind eye to animal welfare practices in the global meat market, and the cruelty against animals involved with beauty, fashion, and cosmetic research. Although this doesn’t mean we should usher in another practice of mistreating animals, because it’s already omnipresent, it does mean we shouldn’t be so quick to dismiss it. In the hierarchy of importance, xenotransplantation is higher than practically every other use of animals in research and trade. As a result, we should ensure a standard of animal welfare is established in these research trials, and then move forward swiftly.
This post highlights the real dangers presented by xenotransplantation and why it is essential to see current issues within the larger picture. If we fail to recognize the concerns raised by zoonotic diseases and animal experimentation, we will find ourselves opening a pandora’s box when these issues emerge and require even more costly solutions. As we have seen with the current pandemic, it only takes one zoonotic disease to change the course of human history and cause immense financial hardship for the global community. Although most repercussions of research affect all people to a certain extent, none do so quite as unavoidably as a pandemic. While xenotransplantation may indeed ameliorate some of the organ shortage concerns, it may delay transplant research that allows us to use stem cells and 3D printing, as L. Syd M. Johnson mentioned. These methods, while they may appear aspirational now, could likely be used more efficiently and ethically than the usage of animals. Also discussed in the post is the need for preventative treatment, which is a critical theme in medicine. In an era of cutting-edge technology and exponential growth of information, we now more than ever need to go back to the basics and focus on maintaining health rather than treating disease. It is clearly preferrable to reduce the incidence of diabetic nephropathy than to see so many patients in dire need of kidneys. Not to mention, disease prevention would minimize the suffering that is caused not just by dying while waiting for an organ, but also by dealing with the complications of receiving one. While some organ failure is inevitable, much of it is not. In the quest to solve this issue, we cannot ignore the foundational problems in our healthcare system by instead pursuing solutions such as xenotransplantation that may well leave us with a larger issue in the end.
This article certainly raised three key issues with xenotransplantation that highlight practical and ethical struggles we face as we attempt to move forward with this technology. Indeed, the idea that the research subjects who “give up” rights to withdraw from surveillance and the right to privacy of those around them (specifically to monitor for zoonotic disease transmission) seems like a violation of some of the most basic research ethics principles. The case of xenotransplantation, however, seems to me to be an extraordinary kind of research, where the possibility of harming not only the subject but those in contract with them is relatively high – safety of close contacts, and in fact as we have now experienced the world, is jeopardized. As a side thought, the research participants haven’t lived long enough for any of them to want to actively withdraw from a study – we are not far enough along yet to have encountered this problem. What is the goal for life expectancy of xenotransplant recipients? The same as with human-to-human transplant? How long will these individuals have to live with “giving up” their rights? Is the trade-off worthwhile?
As we continue xenotransplantation research, I fear Johnson’s objections to this form of transplantation are extremely valid. On a more narrative spectrum, I wonder if this is how we start down the road toward living-as-machines, where humans have interchangeable parts with modified species and machines, and that other life-forms become mere sources of organs on an assembly line grown for our use. Human-to-human organ transplant seems like giving and receiving on more equal footing (tabling all the other issues we have with autotransplant ethics). Have humans not exploited life on earth enough? If we change out enough parts, at what point will we stop being human?
A very well digested article indeed, as it highlights all the pros but also the cons that could easily be dismissed in the light of a hopeful research advancement. I agree most with the closing argument that there should always be an alternative to science and research when there is any question on the endangerment of a living organism’s wellbeing. As seen with the statement, “The ultimate goal of this research is to turn pigs into a ready source of organs for transplant” sounds very much like the “savior sibling debate, where children are conceived for the use of the parents to have them as tools for their other ill child’s medical and health needs. This brings us into the idea that consent is one of the 4Cs of ethics that guide toward what should be legal or what should not.
Here, “potential recipients of xenografts are research subjects whose consent and safety are governed by human subjects research regulations and guidance” is provided for human subjects – so why is this same system of consent not employed for the donor animals as well? I think that if “participation is voluntary . . . and the subject may discontinue participation at any time without penalty or loss of benefits,” then why is this only a right for the humans and not the animals?
There are, for good reason, best practices for animal care and welfare that ensure ethically appropriate environments to meet the animals’ behavioral and physiological needs as mentioned here. I think that those should be implemented even prior to passing study designs in order to minimize the number of ethical compromises, such as this, that occur once the studies are in effect. Once study designs have stipulated and firm regulations across research investigations for the welfare of all living things involved in the investigation, will it become easier to advance in the scientific world. I could not agree more with protecting the consent and voluntary regimens humans generally receive for all living organisms.
I found this article fascinating because it explores a cutting-edge area of research. I hadn’t realize that the pigs used for transplantation come from genetically modified and cloned animals bred under strict infection control measures to prevent any zoonotic viruses. This is just the first step in the transplantation process. Even after a successful transplant, there is still much we don’t know about how these organs affect the human body, especially in highly immunocompromised patients. Recipients and their family members require lifelong surveillance and ongoing monitoring to assess long-term outcomes due to the concerns about the potential for new virus strains and pathogen transmission.
In addition, animal welfare in xenotransplantation is often overshadowed by the focus on human medical advancements. These genetically modified pigs are bred specifically for organ harvesting, raising ethical concerns about their quality of life, potential suffering and the moral implications of using animals in this way. Unlike traditional livestock farming, where there is at least some regulatory oversight regarding humane treatment, the welfare of these animals is less scrutinized in the context of transplantation. As research in this filed progresses, there is growing need for discussions about ethical standards, human treatment, and regulatory oversight to ensure these animals are not subjected to unnecessary suffering.
I appreciate this article’s skeptical view of xenotransplantation, especially as excitement and anticipation for this field has grown in the three years since this article has been posted. I feel that much like many advancements throughout medicine, the harsh criticism or concern surrounding innovations is what will allow these advancements to be implemented in the most ethical manner possible.
However, each of the three considerations this article discusses do not seem to ring the death knell for xenotransplantation, and to me do not suggest that we should necessarily seek alternatives. While I agree that alternatives like preventative medicine, stem cell organ production, or opt-out donor registration systems could contribute to less people dying on the waitlist, if it was so simple to implement these social or scientific changes, I believe we would have done so already. The ethical or societal challenges that come with these alternatives may be just as, if not more, extensive as those with xenotransplantation. And even if xenotransplantation simply acts as a temporary bridge to more long term, ethically sound, solutions, it is a bridge that could save thousands of lives in the meantime.
It is true that there are significant concerns with xenotransplantation’s research and development phase, especially due to the responsibility to ensure that the compassionate use clinical cases are not becoming exploitative or providing patients with a therapeutic misconception. This concern, when combined with the potentially disastrous public health outcomes due to zoonotic pandemics or the scrutiny from public opinion due to animal rights violations, outlines the importance of having the ethics of xenotransplantation further evaluated before innovation in this field exceeds its regulation. As indicated throughout this article, navigating these three principle concerns to xenotransplantation will be critical to this field moving forward if it hopes to become the standard of care for those awaiting an organ for an excessive amount of time.
Now, three years later, in a world where individuals have survived with pig organs and lived infection free for months, the benefits of xenotransplantation often overshadow the necessary ethical considerations when presented to the general public. With flashy articles and headlines indicating the potential benefits of xenotransplantation, it becomes increasingly important that we look back at these foundational concerns and bioethical principles to reflect on how we have worked to consider them thus far, and how we plan to continue to improve upon them going forward.
This article raises an important and complex ethical question about the future of xenotransplantation. As a health care professional and someone passionate about both science and ethics, I find myself torn. On one hand, the idea of genetically modified pig organs saving lives is extraordinary – especially when so many people die waiting for transplants. The more I learn from this course, the more I realize how entangled this potential solution is with risks that reach far beyond the operating room. One point from the article that stood out to me was the conflict between lifelong surveillance of recipients and the right to withdraw from research. As someone who works closely with patients, I can’t imagine asking someone to give up such a fundamental right in the most vulnerable chapter of their life.
I also appreciated the discussion about why pigs have become the preferred research animal. Their biological distance from humans, compared to chimpanzees or monkeys, may lower the risk of zoonotic viruses that could otherwise devastate an immunocompromised patient. Pigs are more commonly bred; the way pigs are treated in this context – isolated, modified, restrained – pushes me to question what we’re willing to compromise for medical progress. Of course, we do need solutions to the organ shortage, but we also need to be honest about the moral trade-offs we’re making.
Dr. Johnson highlights many salient concerns when it comes to the need to balance the dual-sue dilemma posed by the field of xenotransplantation for organ shortages. In light of recent success stories, such as the second xenotransplantation of a pig kidney into a living human recipient at Massachusetts General Hospital, Dr. Johnson’s ethical concerns, while critical, do not seem to signal the end for this solution.
The feedback loop that is formed from the risk of zoonotic disease transmission during transplantation and thus requiring potentially lifelong surveillance seems like a necessary prerequisite for such a procedure. In situations that could become matters of public health, the individual recipient of a xenotransplantation is not the sole stakeholder, which does give credence to calls for continuous monitoring. Dr. Johnson keenly highlights that this conflicts with the ethical principle of autonomy and the right to withdraw at any time without penalty or loss of benefits, which raises a red flag for xenotransplantation. However, how much does this requirement for monitoring practically diverge from conventional organ transplants, and would it be so invasive that patients forewarned during the consenting process would opt out of the procedure? Another point of consideration during the consenting process could include the concept of expanded criteria organs, and how they may be transplanted into patients with chronic conditions. In the event that xenotransplantation became an option for potential recipients, would consistent and prolonged monitoring provide greater measures of beneficence, at some cost of autonomy? As is often the case in healthcare and bioethics, tradeoffs must be made, and for certain patients for whom xenotransplantation becomes a future option, denying this avenue may lean too far in the direction of preventative paternalism, if the zoonotic infectious risks can be practically mitigated with CRISPR and autoimmune therapies.
Another issue that Dr. Johnson did not directly explore, but implicitly raised, is the cost of alternative therapies. Stem cell grown organs, or those made through 3D bioprinting require significant costs in infrastructure development upstream of the actual creation of the organs, which remain products of the future for now. While xenotransplantation, like the recent success at Massachusetts General, shows practical application in the near future, the costs associated with these procedures are not entirely clear. In the ongoing research stage, well-funded institutions are likely footing the bill, but if and when xenotransplantation becomes a viable option for patients, how expensive will they be? Who will bear the costs? In the current model of conventional organ transplantation, the recipient’s insurance usually bears the costs for the surgeries, including for live donations. How does this formula change when it comes to using pigs to grow organs? If the costs at the early stages are too high and insurance companies are unwilling to pay for them, will xenotransplantation become paywalled for wealthy patients? This logistical issue directly interfaces with the bioethical principle of justice, and fair access to organ transplantation. The current model of organ transplantation in the United States heavily relies on Organ Procurement Organizations, which operate regionally, and due to issues in overlapping jurisdictions, rankings of hospitals, accreditation metrics, and other supply-chain related issues, there is an alarmingly high number of organs that go unused and are effectively wasted. With the potential costs associated with xenotransplantation in concert with the costs of organ waste in light of the shortages plaguing healthcare, where is the appropriate balance and focus? Xenotransplantation certainly offers fantastic potential benefits for organs that truly are not being donated at significant rates, however it is paramount that the healthcare community does not look too far ahead to future technologies, in lieu of taking actionable steps now to address organ waste.